|
6/19/2023 0 Comments Co element or comound Other compounds are made from different combinations of atoms, like water… carbon dioxide… and table salt.Īnd that’s it! Nearly everything in this room, in your room, and in the entire universe, is made of: elements with one kind of atom, compounds containing different types of atoms chemically bonded together and mixtures of different elements and compounds together. Iron sulfide, a new substance, has been formed because the iron and sulfur atoms are now chemically bonded together. The iron can no longer be removed by a magnet because it’s bonded to the sulfur. atoms of the two elements are now joined together by chemical bonds. When the iron and sulfur are heated together… We can still separate the elements in the mixture. The different elements are not joined together. This is now a mixture of the elements iron and sulfur. 
When we mix two different pure substances together, like this, it’s a mixture. 
It contains only sulfur atoms, and nothing else, so it is pure. It is made of only one type of atom: iron atoms. That name tells you that you have one carbon (C) atom and one. Some substances, like particles of this iron, contain only one kind of atom. Most simple compounds only have two words in their names. “ Carbon-monoxide-3D-balls” By Benjah-bmm27 – Own work (Public Domain) via Commons WikimediaĢ.Everything we can see and touch, and quite a lot that we can’t as well, is made of tiny particles called atoms. “ Carbon Monoxide.” Encyclopædia Britannica, Encyclopædia Britannica, Inc. “ Cobalt – Element Information, Properties and Uses: Periodic Table.” RSC.Ģ. The key difference between CO and Co is that CO is an inorganic compound consisting of carbon and oxygen atoms, whereas Co is the metal named Cobalt. The below infographic presents the differences between CO and Co in tabular form for side by side comparison.ĬO and Co are two different substances having different chemical and physical properties. So, the key difference between CO and Co is that CO is an inorganic compound consisting of carbon and oxygen atoms, whereas Co is the metal named Cobalt. Cobalt is a chemical element having the chemical symbol Co and atomic number 27. What is the Difference Between CO and Co?ĬO is a colourless, odourless, and tasteless, flammable gas consisting of a carbon atom and an oxygen atom per molecule. Cobalt is a hard, lustrous bluish-gray metal. However, we can produce the free element using the smelting process. Cobalt does not occur as an individual metal on earth’s crust instead, cobalt exists in combination with other elements. 
Furthermore, we can classify it as a transition metal. It is a metal and a d-block element in the periodic table. Iron smelting also produces this toxic gas as a byproduct. We can prepare carbon monoxide using the partial oxidation of carbon-containing compounds such as carbon dioxide. We can identify carbon monoxide as the simplest oxocarbon, and it is isoelectronic with other triple-bonded diatomic species having ten valence electrons, e.g. When considering the chemical structure of carbon monoxide, there is one carbon atom bonded to one oxygen atom through a triple bond containing two pi bonds and one sigma bond. 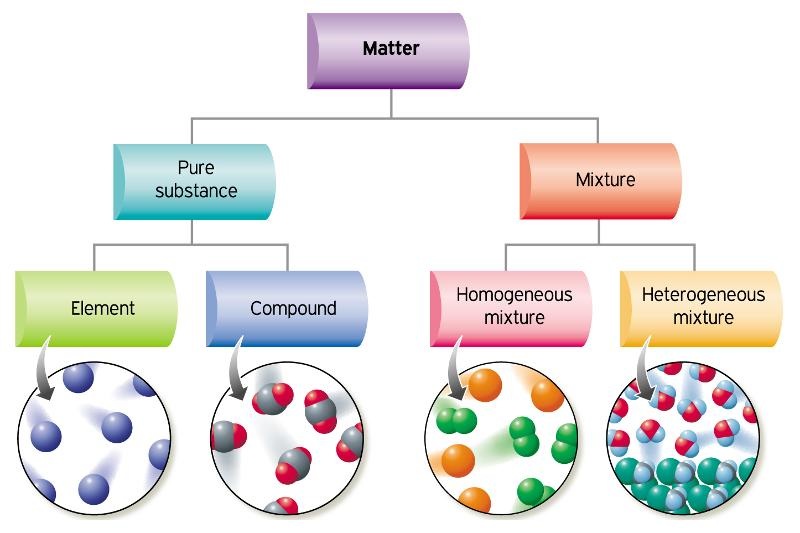
Figure 01: Ball-and-Stick Model of Carbon Monoxide 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
